Address
Room 2301C, 23rd Floor, Building 1, jinghu Commercial center, No, 34, Liangzhuang Street, Eri District, Zhengzhou City, Henan province
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
Address
Room 2301C, 23rd Floor, Building 1, jinghu Commercial center, No, 34, Liangzhuang Street, Eri District, Zhengzhou City, Henan province
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM

This chapter systematically introduces the historical background of vinyl ether polymers, their monomer synthesis (specifically the Reppe reaction), production methods (such as bulk or solution polymerization), and their physical properties (e.g., solubility) and industrial applications. It places particular emphasis on their value in the fields of adhesives, pressure-sensitive adhesives, and coatings, thereby providing a practical reference for materials design and process optimization.
Overview
The general chemical formula for vinyl ether polymers used in the production of adhesives and coatings is as follows.
[−CH2−CH(OR)−]n
R represents a substituent such as an alkyl group (e.g., methyl, ethyl, isobutyl, etc.) or an aryl group;
n is the degree of polymerization, representing the number of repeating units, which determines the polymer’s molecular weight and physical form (ranging, for example, from a viscous oil to a rubber-like solid).
The viscosity of these polymers depends on their molar mass, ranging from viscous oils to rubber-like solids.
Vinyl ethers were first converted into resinous polymers over a century ago. Between 1920 and 1930, the development of Reppe chemistry made them readily accessible, thereby sparking interest within the industrial sector. Subsequently, research into polymerization methods for these compounds began. In 1938, large-scale production of vinyl ether polymers commenced at the Ludwigshafen plant of the former IG Farben (now the primary production site of BASF). After 1940, GAF Corporation began production, followed by Union Carbide, which subsequently withdrew from this field in 1976.
Monomers
Acetylene undergoes the Reppe reaction with alcohols to form vinyl ethers:
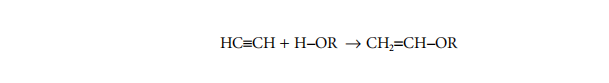
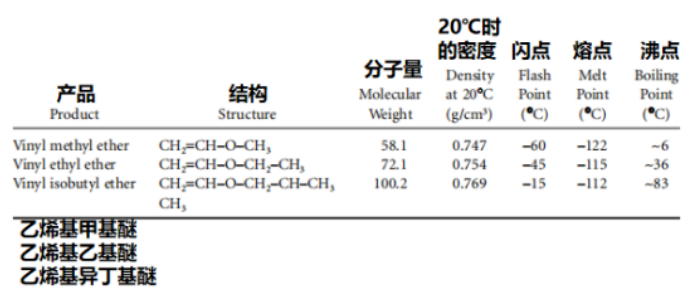
This remains the only industrially significant method for the production of vinyl ethers. The properties of the constituent monomers are listed in Table 1.
Polymer Production
Vinyl ethers can be polymerized in bulk or in solution using either batch or continuous processes. Due to the significant heat of reaction, precise control and specialized equipment are essential. The monomer and initiator are metered continuously into the reactor, and the polymerization reaction commences within a few minutes. Once the reactor is fully charged, it is sealed, and the polymerization continues under pressure. The boiling point of the monomer or solvent determines the rate at which the temperature rises during the reaction. The heat of reaction is removed via a reflux condenser and/or the reactor’s cooling system.
Products on the Market
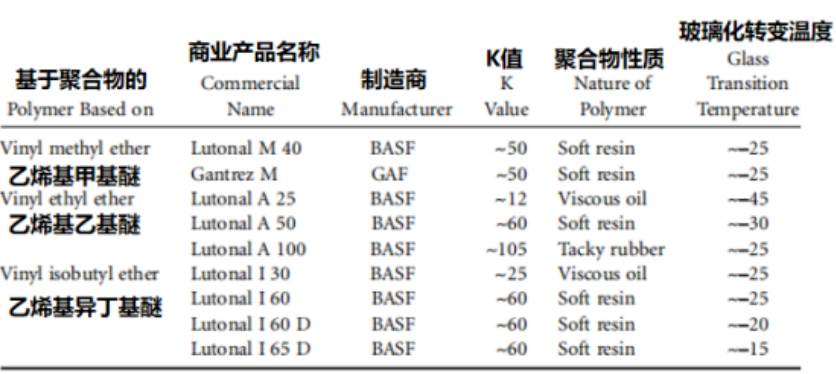
The vinyl ether polymers currently available on the market are listed here. As indicated in the table, the industry typically employs the K-value to characterize the degree of polymerization; this metric serves as a measure of the average molar mass. The physical form in which these polymers are supplied depends on their manufacturing process and intended application requirements; consequently, they may be provided in a solvent-free form or, to facilitate processing, supplied as solutions formulated with common solvents. Lutonal I 60 D and I 65 D are high-viscosity secondary aqueous dispersions with a solids content of 55%; they are synthesized using an 80% solution of Lutonal I 60—dissolved in mineral spirits with a boiling range of 60 to 140°C—and are prepared utilizing different protective colloids.
Feature
The degree of polymerization determines the physical form of polyvinyl ethers (i.e., whether they are viscous oils, viscous plastic resins, or rubber-like substances). Products sold on the market are typically colorless. Occasionally, they may exhibit a slight yellow-to-brown discoloration due to side reactions within the initiator system; however, this generally has no impact on their performance. These polymers possess good resistance to hydrolysis. Products with low to moderate molecular weights (i.e., K 65) may emit a faint odor due to the presence of residual monomers or oligomers.
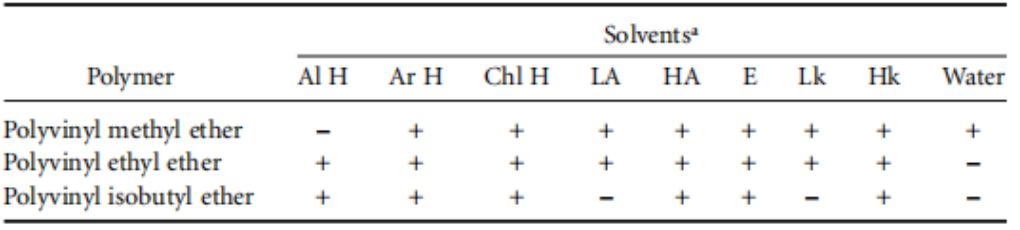
The solubility of vinyl ether polymers is presented in Table 3. Their solubility depends on the alkyl groups. The fact that polyvinyl methyl ether is classified as a water-soluble polymer can be explained by the hydrogen bonding that occurs between the liquid medium—water—and the oxygen atoms within the ether groups. Consequently, the polymer, which is intrinsically insoluble, becomes solvated by the water to form soluble associates. When energy is applied, these hydrogen bonds loosen once again—that is, the polyvinyl methyl ether loses its hydration envelope and subsequently precipitates out. For instance, this phenomenon occurs in a solution of Lutonal M 40 at approximately 28°C. The precipitation point is influenced by the concentration of the aqueous solution, as well as by the presence of any additional solvents.
Application
Vinyl ether polymers constitute a classic class of foundational materials for the production of adhesives and coatings. These materials are primarily utilized in conjunction with other raw materials, or—within the field of pressure-sensitive adhesives—blended with polymers of the same type but differing molecular weights. By incorporating vinyl ether polymers, a range of performance characteristics in base products can be significantly enhanced; these include anchorage strength, adhesion, or tack on difficult-to-bond substrates, as well as resistance to plasticizers and aging.
Key applications in the field of adhesives include blending polyvinyl methyl ether with starch or dextrin for use as labeling adhesives, and mixing medium-molecular-weight polyvinyl ethyl ether with resin solutions for carpet bonding. Secondary dispersions of medium-molecular-weight polyvinyl isobutyl ether, while used in smaller quantities, are of no less importance; they are blended with acrylic dispersions to produce pressure-sensitive adhesives. Furthermore, mixtures of polyvinyl ethyl ether polymers of varying molecular weights are utilized as pressure-sensitive adhesives within the pharmaceutical sector.
In the field of surface coatings, polyethylene methyl ether and medium-molecular-weight polyethylene ethyl ether are frequently formulated in combination with nitrocellulose, chlorinated binders, and styrene copolymers. These formulations are utilized for coating flexible substrates—such as metal foils, plastics, films, and paper—as well as for the preparation of antifouling coatings.
We can provide Vinyl Ether Polymer list:
CAS:107-25-5 Name:Methyl vinyl ether
CAS:764-47-6 Name:n-propyl vinyl ether
CAS:111-34-2 Name:n-butyl vinyl ether
CAS:109-53-5 Name:Isobutyl vinyl ether
CAS:2182-55-0 Name:Cyclohexyl vinyl ether
CAS:17832-28-9 Name:4-Hydroxybutyl vinyl ether
CAS:103-44-6 Name:2-Ethylhexyl vinyl ether
CAS:929-62-4 Name:n-Octyl vinyl ether
CAS:765-14-0 Name:n-Dodecyl vinyl ether
CAS:930-02-9 Name:n-Octadecyl vinyl ether
CAS:3891-33-6 Name:1,4-Butanediol divinyl ether
CAS:764-48-7 Name:Ethylene glycol vinyl ether
CAS:764-78-3 Name:Ethylene glycol divinyl ether
CAS:929-37-3 Name:Diethylene glycol monovinyl ether
CAS:764-99-8 Name:Diethylene glycol divinyl ether
CAS:765-12-8 Name:Triethyleneglycol divinyl ether
CAS:9003-09-2 Name:Poly(methyl vinyl ether), LUT-M20 & M40
CAS:25104-37-4 Name:Poly(ethyl vinyl ether), LUT-A25 & A50
CAS:9003-44-5 Name:Poly(isobutyl vinyl ether), LUT-130/145/160
CAS:92680-80-3 Name:Poly(n-butyl vinyl ether), LUT-N65; Vinilin
CAS:25154-85-2 Name:MP25/MP35/MP45Copolymer of vinyl chloride and vinyl isobutyl ether,
MP25/MP35/MP45