Address
Room 2301C, 23rd Floor, Building 1, jinghu Commercial center, No, 34, Liangzhuang Street, Eri District, Zhengzhou City, Henan province
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
Address
Room 2301C, 23rd Floor, Building 1, jinghu Commercial center, No, 34, Liangzhuang Street, Eri District, Zhengzhou City, Henan province
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM

Incorporating adhesion promoters into a coating formulation—thereby facilitating reactions between the substrate and the coating film through hydrogen bonding or interactions involving hydroxyl or carboxyl groups—constitutes the most effective method for enhancing adhesion.
Common adhesion promoters include:
(1) Silane coupling agents
(2) Phosphate ester compounds
(3) Titanate or zirconate coupling agents
(4) Polymeric organic resins
(5) Chlorinated polyolefins
The following section provides a brief overview of the underlying mechanisms of these five types of adhesion promoters to facilitate an understanding of their applications and characteristics.

I. Silane Coupling Agents
A coupling agent is a molecule featuring two distinct functional groups—one organophilic and the other inorganic-philic—located at its opposing ends. It serves to bridge materials with disparate chemical structures and widely differing affinities at their interface, thereby enhancing the bonding between the resin and inorganic substrates, pigments, or fillers. As illustrated below:
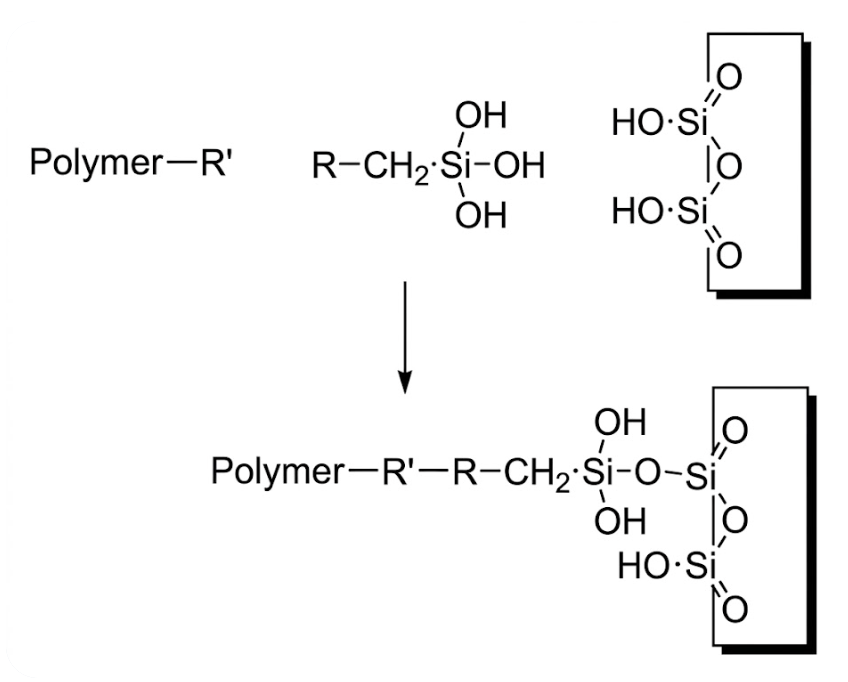
The general formula for silane coupling agents is R-(CH₂)n-Si-X(4-n), wherein R represents a non-hydrolyzable organic functional group capable of reacting with polymeric materials—such as hydroxyl, vinyl, amino, epoxy, or methacrylate groups. X represents a hydrolyzable functional group; upon contact with moisture adsorbed on the surface of inorganic materials, it undergoes hydrolysis and exhibits favorable reactivity with the inorganic surface. Typical X groups include methoxy (OCH₃) and ethoxy (OC₂H₅) groups.
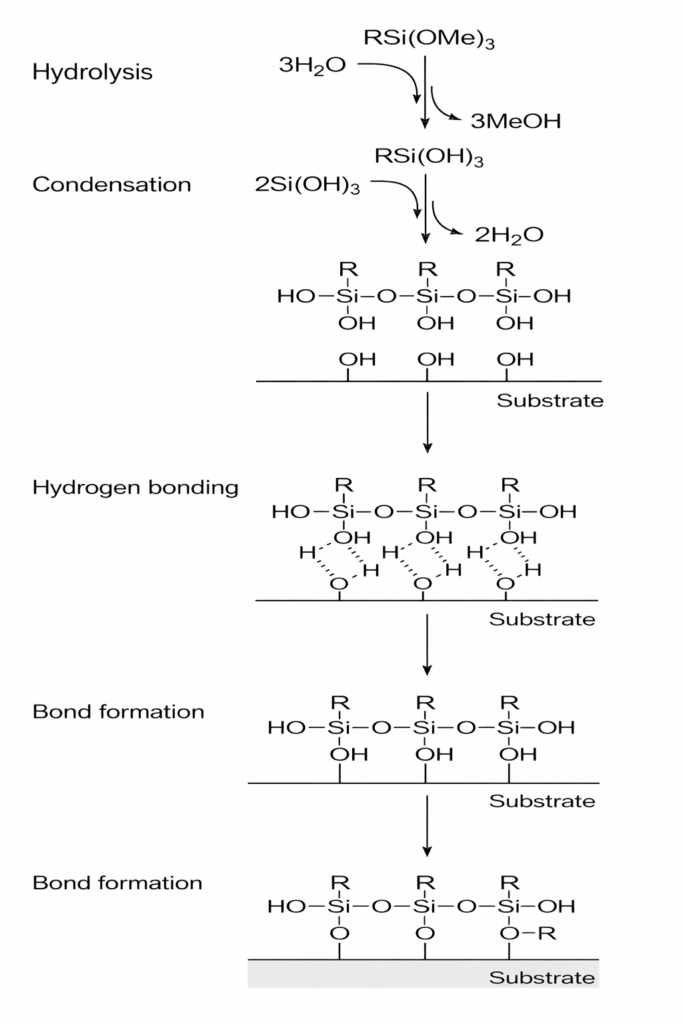
One of the most common examples of incorporating silane coupling agents into coatings involves their application to glass substrates (such as wine bottles, cosmetic containers, etc.). Following spray application, the silane coupling agent encounters moisture present on the inorganic glass surface; it subsequently hydrolyzes into silanols, which then form hydrogen bonds or undergo condensation reactions with the hydroxyl (OH) groups on the glass surface to create covalent Si-O-M bonds. Simultaneously, the organic functional group at the other end of the molecule reacts with the resin matrix. The reaction mechanism for amino silanes when utilized as adhesion promoters is illustrated as follows:
II. Phosphate Compounds
The structure of phosphate ester compounds incorporates various organic functional groups, as well as phosphate groups capable of reacting with inorganic metal substrates; their chemical structure is as follows:
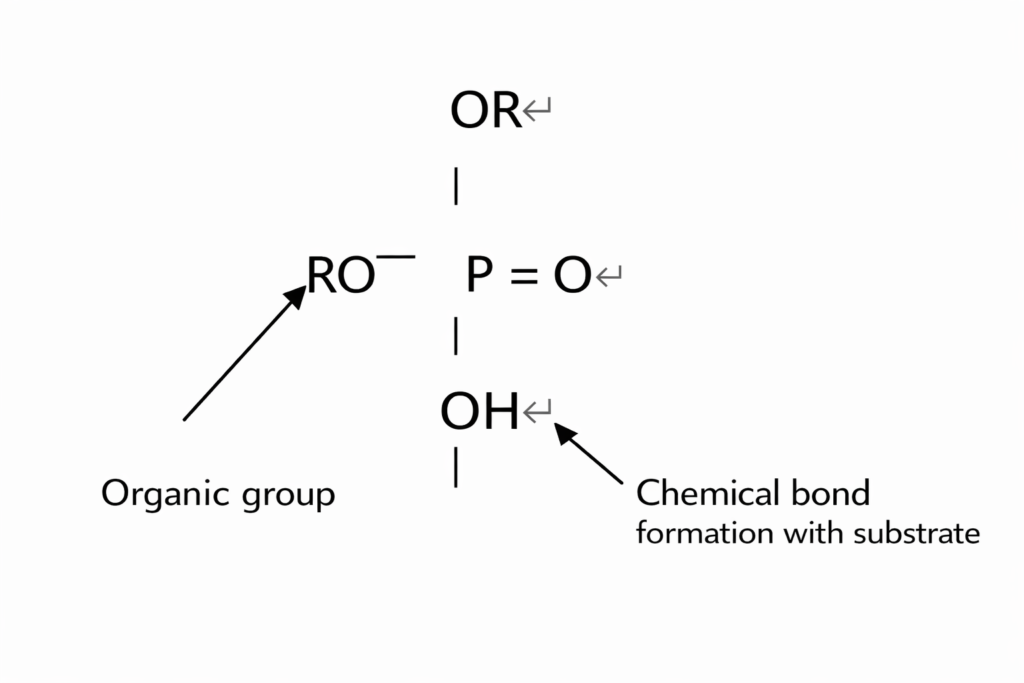
Epoxy phosphates constitute the most significant class of phosphate esters; under baking conditions, they demonstrate exceptional adhesion-promoting properties on metal substrates. For further details, please refer to:
Synthesis and Application of Novel Water-based Epoxy Phosphates
III. Titanates or Zirconates
The molecular structure of titanates or zirconates is illustrated in the figure below. The organic segment of the molecule is capable of undergoing a hydrolysis reaction with moisture present on a metal surface to form chemical bonds; simultaneously, the other end reacts with the functional groups of the coating resin, thereby functioning as an adhesion promoter.
(RO)(4-n)-Ti-(OX-R-Y)n
n = 1–3
RO: Hydrolyzable short-chain alkoxy group
OX: –COOH, –SO₃H, Phosphate group
R: Long-chain alkyl group
Y: –OH, –NH₂, Epoxy group
IV. Polymeric Polymers
Products of this type are characterized by a high density of organic functional groups. Coupled with a specialized structural design, they typically exhibit a high hydroxyl value, a high acid value, and exceptional flexibility—qualities that serve to enhance the adhesion of coatings to metal substrates, inorganic substrates, or certain plastic components. Furthermore, certain polymer-based adhesion promoters within this category can also improve “silver-binding” properties (the ability to anchor aluminum flakes) and alcohol resistance in aluminum-pigmented coatings.
When these adhesion promoters are incorporated into a coating formulation, they function by rapidly migrating via ionic charge and effectively wetting and penetrating the substrate surface following application. During high-temperature baking, they form chemical bonds with the metal substrate—specifically through hydrogen bonding—while simultaneously reacting with amino-based crosslinking agents, thereby achieving a significant enhancement in adhesion.
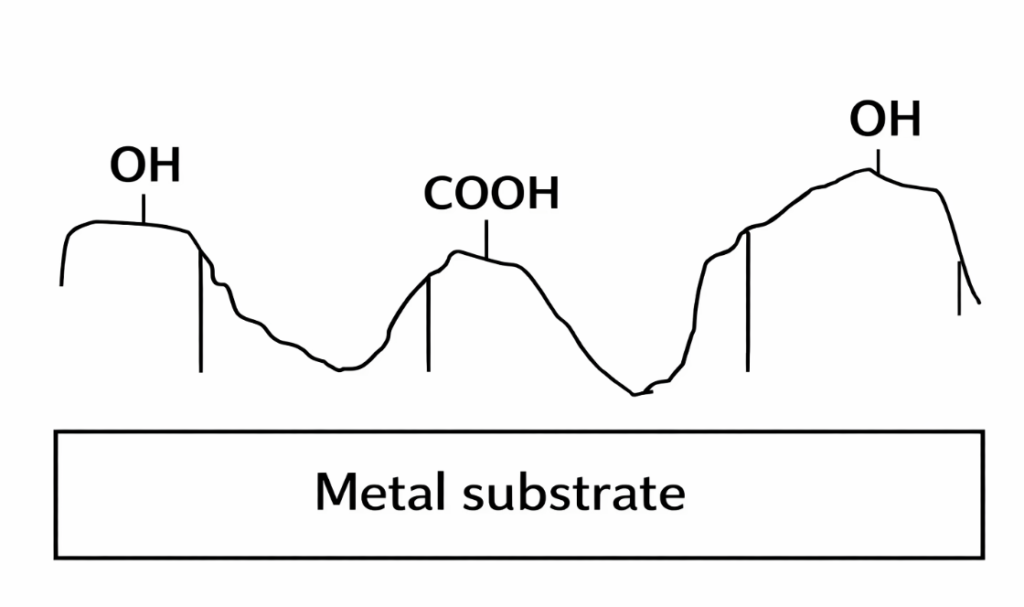
V. Chlorinated Polyolefins
Polypropylene (PP) substrates are characterized by high crystallinity, low polarity, and low surface energy, making it difficult for conventional coatings to adhere to them. Consequently, chlorinated polyolefins—materials possessing similar polarity—are employed to enhance adhesion. Through processes involving wetting, penetration, diffusion, and molecular motion, the chlorinated polyolefin polymers intermix with the substrate, thereby improving adhesion strength. The structural formula of chlorinated polyolefins is illustrated below; the modifying groups—such as chlorine, acrylic acid, or maleic anhydride—serve to improve compatibility with the resin system as well as interlayer adhesion. For a detailed explanation of the specific mechanism, please refer to:
PP Substrate Adhesion Promoters
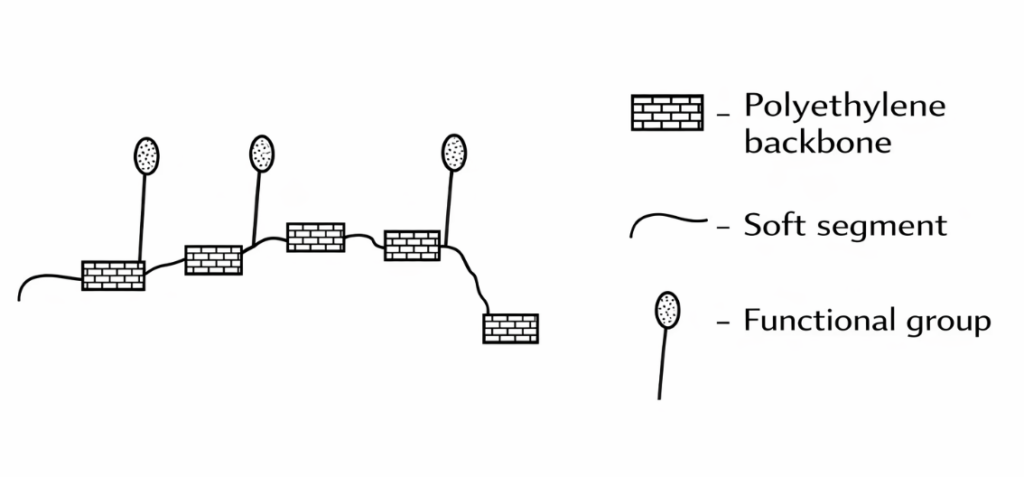
When selecting an appropriate adhesion promoter, it is essential to consider the type of coating, the nature of the substrate, and the characteristics of the application environment. For instance, for water-based coatings, a water-based adhesion promoter should be selected; conversely, for oil-based coatings, an oil-based adhesion promoter is required. The typical addition level for an adhesion promoter ranges from 0.5% to 3% of the total coating volume; it is generally added during the early stages of the coating formulation process to ensure its uniform distribution throughout the mixture.