Address
Room 2301C, 23rd Floor, Building 1, jinghu Commercial center, No, 34, Liangzhuang Street, Eri District, Zhengzhou City, Henan province
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
Address
Room 2301C, 23rd Floor, Building 1, jinghu Commercial center, No, 34, Liangzhuang Street, Eri District, Zhengzhou City, Henan province
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM

In the field of electrochemical research, Nafion is an indispensable subject. However, many students frequently encounter misconceptions during their experiments; they often mispronounce or misname it—some even mistakenly refer to it as “naphthol”—or they may understand what it is without fully grasping the underlying principles. To address this issue, we have compiled this summary covering the essentials regarding Nafion, Nafion membranes, and Nafion solutions.
What is Nafion?
According to DuPont’s official website, the full name of Nafion is perfluorosulfonic acid polymer. It was developed in the late 1960s by Dr. Walther Grot through the modification of Teflon material, and it stands as the very first synthetic polymer ever created to possess ionic properties. Its molecular structure is illustrated in Figure 1; internally, the chain ends of its hydrophobic domains terminate in hydrophilic sulfonic acid groups. These groups exhibit excellent selectivity toward cations; based on this characteristic, Nafion is frequently utilized as a proton exchange membrane.
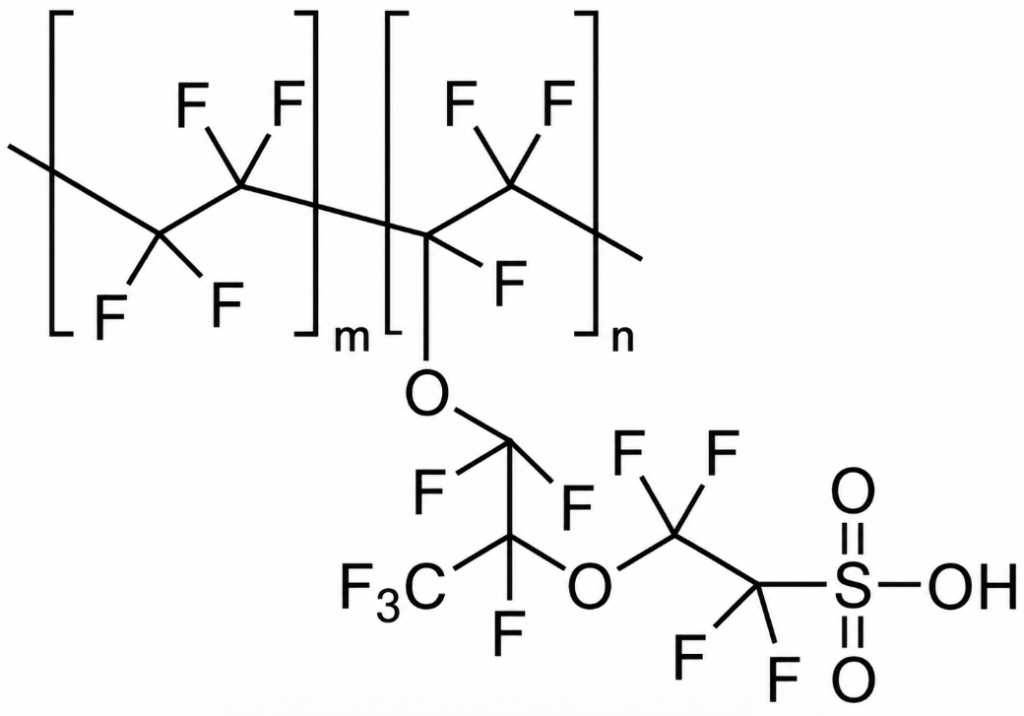
Nafion combines many of the physicochemical properties of polytetrafluoroethylene (PTFE) with its own inherent ionic characteristics, resulting in a material that possesses several unique properties:
Nafion, Nafion Membranes, and Nafion Solutions
Given Nafion’s numerous excellent properties, its utilization has led to the development of many derivative products—among which Nafion membranes and Nafion solutions are two examples.
Nafion membrane
Nafion possesses excellent ionic conductivity; consequently, it is frequently employed in electrocatalysis as a cation-exchange membrane—primarily for protons. It is an essential component in the investigation of various electrochemical reactions, such as the Oxygen Evolution Reaction (OER) and the Hydrogen Evolution Reaction (HER)—hence the term “Nafion membrane.”
It is worth noting that Nafion membranes are categorized into various models based on specific differences in properties, such as mechanical strength and conductivity. The most widely used variant is Nafion 117; however, thinner alternatives—such as Nafion 212 and Nafion 115—are also available, allowing users to select the most suitable product based on the specific requirements of their application.

Prior to use, Nafion membranes typically require certain pretreatment steps. The standard procedure is as follows:
First, treat the membrane with a 5% (by mass) hydrogen peroxide solution at 80°C for one hour, followed by soaking in deionized water for 30 minutes. Next, boil the membrane in a 5% (by mass) dilute sulfuric acid solution at 80°C for one hour. Finally, soak it in deionized water for another 30 minutes; the membrane is then ready for use. Alternatively—depending on the user’s established practices—one may simply soak the membrane in the electrolyte solution before use.
The rationale behind this pretreatment lies in the fact that Nafion membranes possess a certain thickness; consequently, it takes some time for them to fully engage in ion exchange after being immersed in an electrolyte solution. Therefore, to ensure that the ion exchange process proceeds without interference, thorough soaking is essential. Additionally, the use of hydrogen peroxide helps remove any cations that may have adsorbed onto the membrane’s surface during its preparation and handling.
Nafion 5% Membrane Solution

Nafion solution is primarily utilized to employ Nafion as an electrode modifier. This necessitates a discussion of chemically modified electrodes.
This represents an emerging field that developed in the 1970s—and remains one of the most active frontiers in electrochemistry and electroanalytical chemistry today. In 1989, the IUPAC Commission on Electroanalytical Chemistry formally named and defined chemically modified electrodes, stating: A chemically modified electrode is an electrode fabricated from a conductor or semiconductor, featuring a thin surface coating of chemical substances—whether monomolecular, multimolecular, ionic, or polymeric—that manifests the chemical, electrochemical, or optical properties of this modifying film through Faradayic reactions.
In electrocatalytic processes utilizing glassy carbon or carbon electrodes, a common challenge is the tendency for the catalyst to detach from the electrode surface. Since Nafion is a polymer possessing inherent viscosity—along with excellent electrical conductivity—it is ideally suited to address this issue by leveraging these specific properties. Consequently, in numerous research endeavors—particularly during the preparation of catalysts for reactions such as the Oxygen Reduction Reaction (ORR) and Oxygen Evolution Reaction (OER)—a specific quantity of Nafion solution is routinely incorporated.
However, several factors regarding the addition of Nafion solution warrant careful consideration. Specifically, the sequence of addition is critical. Generally, there are two primary methods for introducing Nafion: first, it can be mixed directly into the catalyst “ink,” thereby forming a composite modification layer alongside the catalyst; second, the catalyst ink can be deposited onto the electrode surface first, followed by the application of a thin protective layer of Nafion solution over the resulting film. From the perspective of preventing catalyst detachment, the former method is undoubtedly more advantageous; however, when considering the potential for Nafion to interfere with the actual reaction kinetics, the latter approach appears to be preferable. Furthermore, some contemporary studies opt to forgo Nafion as a binder or cross-linking agent entirely, choosing instead to employ alternative substances—such as isopropyl alcohol—depending on the specific requirements of the system under investigation.
Additionally, the optimal quantity of Nafion to be added must be carefully determined. Evidently, the principle of “too much of a good thing” applies here: while the inclusion of Nafion offers numerous benefits, applying an excessive amount to the catalyst surface results in the formation of a polymer film that inevitably hinders direct contact between the catalyst surface and the electrolyte solvent molecules. This interference can potentially disrupt the electrocatalytic surface reaction processes. Moreover, an excessive application of Nafion may also lead to an undesirable increase in the internal resistance of the electrochemical cell.
If you would like to learn more about Nafion, please do not hesitate to contact us. If you wish to purchase Nafion solutions or membranes, please reach out to contact@quartz-chem.com; we offer the best pricing and worldwide delivery services.