Address
Room 2301C, 23rd Floor, Building 1, jinghu Commercial center, No, 34, Liangzhuang Street, Eri District, Zhengzhou City, Henan province
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
Address
Room 2301C, 23rd Floor, Building 1, jinghu Commercial center, No, 34, Liangzhuang Street, Eri District, Zhengzhou City, Henan province
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM

With the vigorous development of water-based industrial coatings, the consumption of water-based alkyd resins and water-based epoxy esters is steadily increasing. This paper attempts to summarize the application of commonly used metal catalysts.
Drier Types
Driers can be classified into three types: oxidative, polymerizing, and auxiliary:
1.Oxidative Type (Surface Drying): Co, Mn, Ce, Fe
Co — The most active and potent oxidative drier.
Ce, Fe — Baking-type driers.
Mn — A dual-functional drier exhibiting both oxidative and polymerizing properties.
2.Polymerizing Type (Through Drying): Pb, Zr, Re
Pb: Traditional polymerizing drier.
Zr: Lead-free substitute.
Re: Used in low-temperature and high-humidity environments.
3.Auxiliary Type (Co-driers): Ca, Zn
Ca: Enhances both surface drying and through drying.
Zn: Improves surface drying properties and prevents wrinkling.

The Mechanism of Action of Driers
The primary reaction mechanisms of driers (specifically primary driers) are as follows:
(1) Dehydrogenation of the α-methylene group of double bonds;
(2) Promotion of the decomposition of generated peroxides, thereby accelerating resin polymerization.
Taking cobalt salts as an example, the reaction process proceeds as follows:
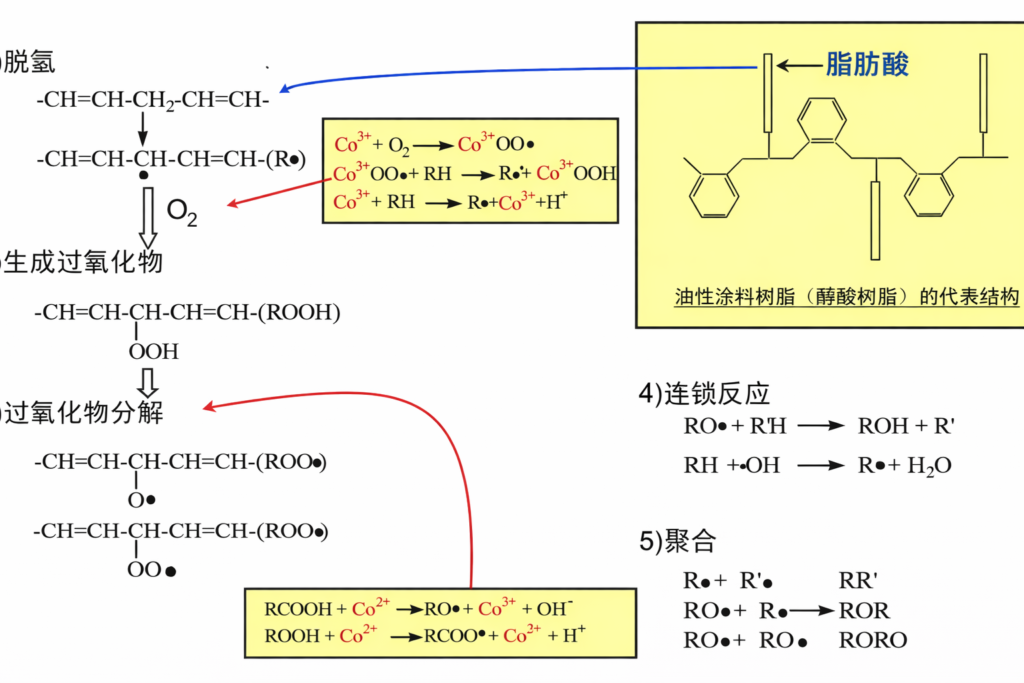
Primary driers possess a strong capacity for oxygen transfer, enabling paint coatings to achieve rapid surface drying; however, they tend to form a sealed surface layer, which can hinder the drying of the underlying layers. The primary driers principally include Co, Mn, V, and Ce.
Auxiliary driers—also known as through-driers—typically consist of metal soaps existing in an oxidized state. They are generally employed in conjunction with primary driers to enhance the latter’s catalytic drying efficiency, thereby ensuring that the polymerization process proceeds synchronously throughout both the surface and interior layers of the coating. The auxiliary driers principally include Ca, Pb, Zr, Zn, Ba, and Sr.

Usage and Precautions for Metal Salts
Adding amounts reaching 0.08% poses a risk; excessive use can lead to wrinkling, loss of gloss, embrittlement, discoloration, and skinning.
Cobalt naphthenate can also be used in amino-based baking enamels (at 0.005–0.02%); it enhances hardness and, compared to Fe and Mn, is less prone to discoloration—though its hardness and toughness are inferior to the latter.
Cobalt soaps typically appear blue-violet, while the higher-valence cobalt state appears green. During the mixing process with paint, peroxides react with the cobalt soap to produce a green color; generally, cobalt soaps exhibit their catalytic activity only when in this higher-valence state. They are typically used at a metal concentration of 6–12%.
At concentrations of 0.5–1.0%, it causes “leafing” aluminum pigments to lose their floating effect, resulting in a grayish appearance.
It acts as a grinding aid and helps reduce viscosity.
It possesses anti-corrosive properties.
Typical usage concentrations are 24–36%.
Its drying catalytic activity is weaker than that of Co, but it provides excellent “through-drying” (bottom-drying) properties.
When used in amino-based baking enamels (at 0.005–0.02%), it enhances both hardness and toughness.
It is prone to causing discoloration, wrinkling, and whitening.
When used in combination with cobalt and lead, it enhances their drying efficiency, balances surface drying with through-drying, and helps eliminate wrinkling.
Typical usage concentrations are: 4%, 6%, and 10%.
It acts as a wetting agent and aids in pigment dispersion.
Typical usage concentrations are: 6%, 8%, 16%, and 18%.
They exhibit no activity at room temperature; however, at high temperatures, they demonstrate strong polymerization-promoting drying activity, imparting maximum hardness and toughness to the paint film. They are frequently used in thermosetting coatings.
Iron-based driers have a dark color, and their drying reaction temperature typically exceeds 110°C.
Their required dosage is approximately half that of lead-based driers, and they do not require the addition of co-driers such as calcium or zinc. 0.5% lead soap is equivalent to 0.33% cerium soap.
When used in combination with cobalt soap (at a ratio of Co: 0.01–0.02% and Re: 0.05–0.1%), it serves as a substitute for lead catalysts. It imparts hardness, water resistance, and weather resistance to the coating, while also improving storage stability by mitigating issues such as hazing, thickening, and gelation.
Usage concentrations: 24%, 18%, 12%, 6%. This acts as a coordination-type polymerization drier; it forms coordination complexes of higher molecular weight by chelating with hydroxyl (OH) groups or other polar functional groups, thereby becoming an integral component of the cured paint film itself.
It exerts a strong synergistic catalytic effect on other driers, shortens drying times more effectively than lead-based driers, and significantly enhances the gloss of the paint film.
These metals possess anti-mold and anti-corrosive properties and are frequently utilized as antifouling agents in marine bottom paints.

Traditional organic acid soap driers—such as those based on Co, Pb, Mn, Zn, and Ca—suffer from various drawbacks: some are deeply colored, some are expensive, and others are toxic. In recent years, however, numerous rare-earth-based driers have emerged, and the associated research and technical methodologies are currently undergoing gradual refinement.