Address
Room 2301C, 23rd Floor, Building 1, jinghu Commercial center, No, 34, Liangzhuang Street, Eri District, Zhengzhou City, Henan province
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
Address
Room 2301C, 23rd Floor, Building 1, jinghu Commercial center, No, 34, Liangzhuang Street, Eri District, Zhengzhou City, Henan province
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM

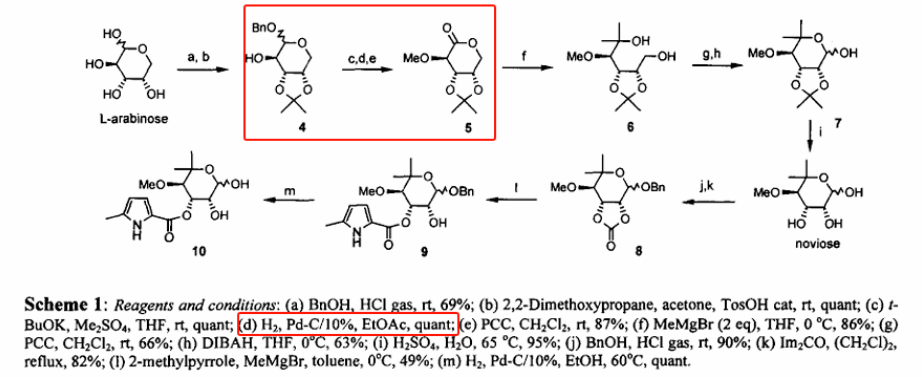
Palladium-on-carbon catalytic hydrogenation is one of the most frequently employed reactions in organic synthesis, widely utilized for the removal of protecting groups containing readily hydrogenolyzable structures—such as benzyl (Bn), benzyloxycarbonyl (Cbz), allyl, allyloxycarbonyl (Alloc), p-methoxybenzyl (PMB), and azido groups.
Although seemingly simple, this step often constitutes the primary bottleneck. The issue of palladium catalyst instability remains a persistent challenge; while previous studies have introduced catalyst pretreatment methods—such as the use of DMF/H₂O and HCl solutions to suppress side reactions—performance discrepancies among carbon-supported palladium catalysts from different sources have yet to be fully elucidated.
For instance, take a synthesis reaction that Caizi performed previously: it appeared quite straightforward—and the literature even reported a quantitative yield—yet we were consistently unable to reproduce the results using domestically produced palladium-on-carbon. Ultimately, the issue was resolved only after switching to imported palladium-on-carbon.
So, why is that?
The article below might provide the answer!
In 2021, Chen Shuai from the Institute of Coal Chemistry at the Chinese Academy of Sciences, together with Stefan Oscarson and colleagues from University College Dublin, published an article in OPRD confirming that performance variations in carbon-supported palladium catalysts primarily stem from inconsistencies in their physicochemical properties—specifically, characteristics such as palladium particle size, distribution uniformity, and oxidation state, which are difficult to control and predict. Furthermore, in practical applications, catalyst selectivity is often compromised by side reactions (such as the saturation of aromatic protecting groups), which in turn affects the purity of the target product and the complexity of its separation.
During the course of the study, researchers first synthesized a decasaccharide structure featuring a branched tertiary architecture, comprising a total of 25 groups—specifically benzyl ether, naphthylmethyl ether, and azide moieties—awaiting reduction. Subsequently, they screened three palladium catalysts sourced from two different reagent suppliers to evaluate the performance differences among various palladium-on-carbon catalysts.
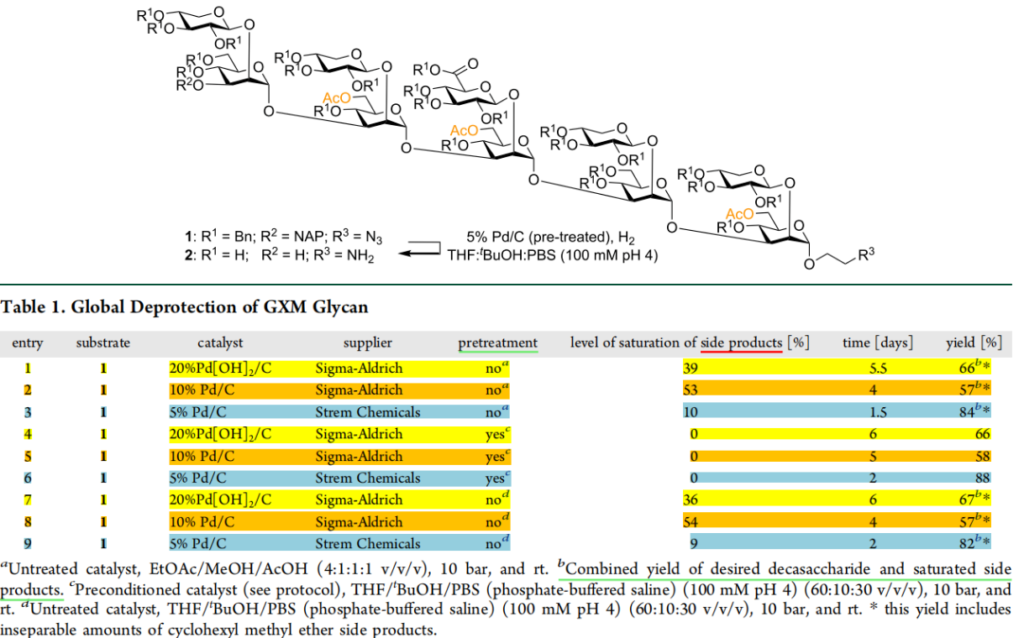
The results indicate that:
The 5% Pd/C catalyst (Strem Chemicals) exhibited optimal performance, characterized by the shortest reaction time (1.5–2 days), the highest yield (82–84%), and the lowest degree of aromatic protecting group saturation (10%). Furthermore, through a pretreatment method (DMF:H2O, 37% HCl), the formation of byproducts resulting from the saturation of aromatic protecting groups was avoided, thereby preserving the desired 6-O-acetylation pattern.
This clearly demonstrates that the catalytic performance of the palladium-on-carbon catalyst is independent of its palladium content!
Subsequently, the researchers employed a variety of characterization techniques to distinguish the fundamental nature of the relevant palladium catalysts:
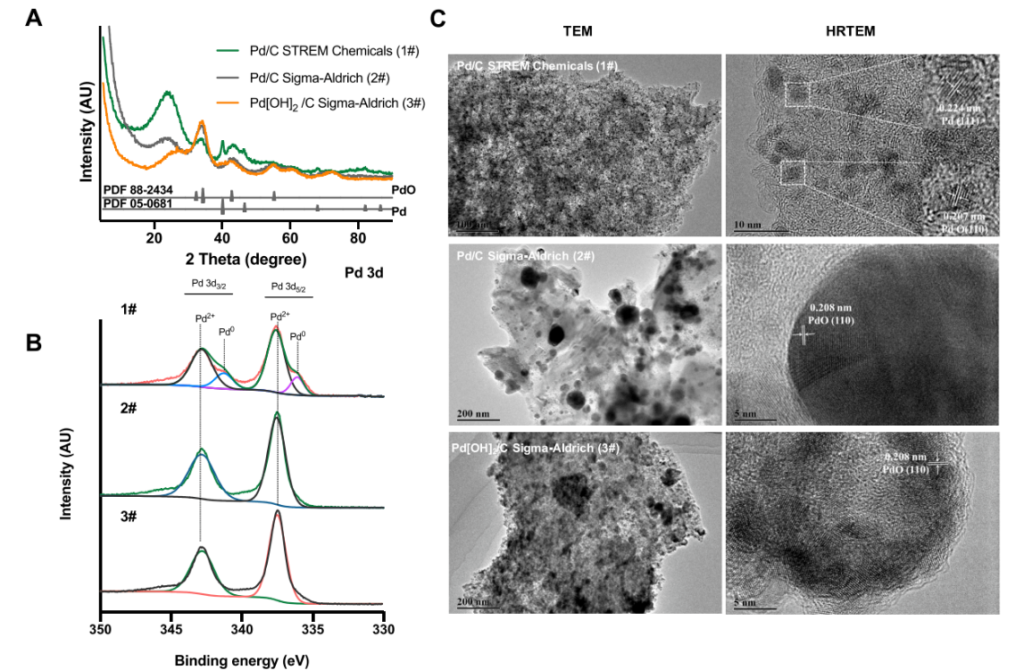
① XRD analysis of the most effective catalyst (5% Pd/C, Strem Chemicals) revealed distinct crystalline peaks corresponding to PdO at 2θ values of 33.3°, 34.4°, 42.9°, and 55.3°; simultaneously, peaks corresponding to metallic Pd were observed, indicating that the catalyst comprises both PdO and Pd⁰. Other lower-quality catalysts consisted primarily of large PdO particles, resulting in longer reaction times and lower catalytic activity.
② Transmission electron microscopy (TEM) analysis revealed that the highly efficient catalyst (5% Pd/C, Strem Chemicals) featured Pd and PdO nanoparticles uniformly dispersed across the carbon support, with an average particle size of approximately 4 nm. In contrast, the lower-quality catalysts exhibited severe particle aggregation and a non-uniform size distribution—characteristics detrimental to the catalytic process.
③ X-ray photoelectron spectroscopy (XPS) studies revealed that, although all Pd-C catalysts contain both Pd²⁺ and Pd⁰, a higher proportion of Pd⁰ leads to superior catalytic performance.
Regarding the issue of catalyst recovery and recycling:
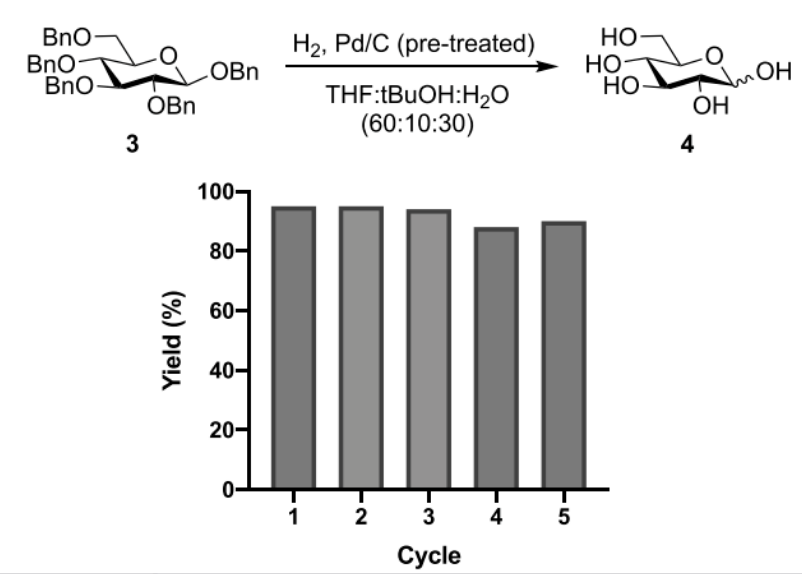
Using fully benzylated glucoside 3 as a model substrate, researchers observed that the Pd/C catalyst—after being recovered via centrifugation—showed no signs of deactivation following five reaction cycles, with yields ranging from 95% to 88%. The coexistence of Pd and PdO persisted, and the small particles remained uniformly dispersed, thereby demonstrating that the catalyst retained its activity even after multiple cycles.
Pd/C Pretreatment Procedure: 500 mg of Pd/C was added to a 10 mL round-bottom flask and suspended in a DMF/H2O mixture (1 mL, 80:20 v/v). 200 μL of HCl (ACS reagent, 37%, pH 2–3) was added, and the mixture was stirred for 20 minutes under a hydrogen atmosphere. The treated Pd/C catalyst was subsequently recovered by filtration and used directly in the hydrogenolysis reaction.