Address
Room 2301C, 23rd Floor, Building 1, jinghu Commercial center, No, 34, Liangzhuang Street, Eri District, Zhengzhou City, Henan province
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
Address
Room 2301C, 23rd Floor, Building 1, jinghu Commercial center, No, 34, Liangzhuang Street, Eri District, Zhengzhou City, Henan province
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
Hydrogen energy is a highly efficient, clean, and safe secondary energy source, and its development and utilization will be an important direction for global energy strategic transformation. Hydrogen has a calorific value of up to 140 MJ/kg, which is 3 to 4 times that of the same weight of fossil fuels. Through fuel cells, it can achieve a comprehensive conversion efficiency of 80 to 90%, or even exceeding 90%. Under the “dual carbon” target (carbon reduction and emission reduction), the hydrogen energy industry is developing rapidly, the fuel cell industry is ushering in new opportunities, and related technologies and materials are receiving unprecedented attention.
As a key material in the hydrogen energy industry chain, proton exchange membranes (PEMs) are essentially ion-selective permeable membranes. Originally used in pure water production, seawater desalination, chemical catalysis, and the chlor-alkali industry, PEMs have been widely applied in recent years with the development of new energy industries and technologies, including water electrolysis for hydrogen production, vanadium redox flow batteries, and catalysis. Currently, the most commonly used perfluorosulfonic acid proton exchange membranes are prepared from perfluorosulfonic acid ion exchange resin through melt extrusion or solution casting. They are typically colorless and transparent films with an average molecular weight between 10⁵ and 10⁶.
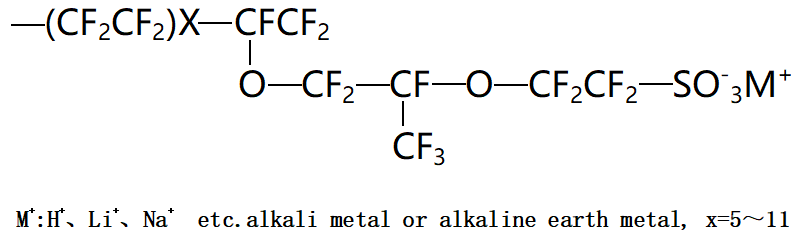
The image shows the chemical structure of Chemours’ PFSA resin, which mainly consists of two parts: the resin backbone is a fluorocarbon linear framework similar to polytetrafluoroethylene (PTFE), providing the membrane with thermal stability, chemical stability, and high mechanical strength; the side chains are perfluoroether structures with sulfonic acid or sulfonyl fluoride groups, providing the membrane’s ion exchange capacity. Different manufacturers’ PFSAs have different side chain structures, such as 3M, Solvay, AGC, and Asahi Kasei, with different numbers of carbon atoms in the side chains compared to Chemours. Perfluorosulfonic acid resins are generally prepared by copolymerizing ether monomers containing sulfonyl fluoride groups with binary or multi-component monomers such as tetrafluoroethylene, hexafluoropropylene, perfluoroalkyl ethers, and perfluorocyclic monomers, and the structures of the sulfonyl fluoride monomers also vary. The composition and ratio of monomers have a significant impact on the chain structure, leading to large differences in molecular weight, molecular weight distribution, sequence composition, and uniformity between different chains; different initiators also affect the end groups, molecular weight, and chain structure; the choice of polymerization method is also very important, as different polymerization methods result in polymers with significantly different properties.
What are the uses of perfluorosulfonic acid resins?
Perfluorosulfonic acid resin is one of the most crucial raw materials for proton exchange membrane fuel cells (PEMFCs). On the one hand, the technology required for the membrane is highly sophisticated, and only a very few companies have mastered this production process. On the other hand, because tetrafluoroethylene, the raw material, is a highly hazardous chemical, and the synthesis from perfluorovinyl ether monomers with sulfonyl fluoride groups is difficult, the production of perfluorosulfonic acid resin for proton exchange membranes has high technical barriers. It requires sophisticated technology in raw material selection and synthesis processes, and generally only fluorochemical companies with substantial R&D capabilities and a complete fluorochemical industry chain possess the production capacity.
Perfluorosulfonic acid ion exchange membranes have a wide range of applications, primarily in water treatment, diffusion dialysis, concentration and purification, ion exchange membranes in the chlor-alkali industry, ion exchange membranes in the fuel cell industry, the nuclear energy industry, and analytical processes. Among these applications, the chlor-alkali industry and the rapidly developing fuel cell industry are currently the most important.
Ion exchange membranes are a crucial component of chlor-alkali production via ion-exchange membrane electrolysis. Their application in the chlor-alkali industry has a long history. While the use of ion-exchange membranes as diaphragms in chlor-alkali electrolyzers was proposed in the 1950s, it wasn’t until the mid-1960s that DuPont developed the chemically stable perfluorosulfonic acid ion exchange membrane, Nafion®. Since then, industrial ion exchange membrane technology has been widely adopted. Since 1970, ion exchange membranes have been extensively used in the chlor-alkali industry, enabling the direct production of NaOH solutions with a purity of over 30% without concentration. These solutions have low impurity content and can be used directly without further purification. Compared to traditional diaphragm and mercury electrolysis processes, this improves production efficiency, reduces environmental pollution, and saves energy costs.
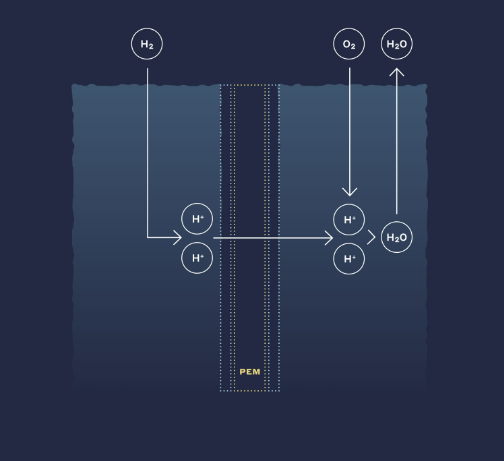
A fuel cell (Proton Electrolyte Membrane Fuel Cell, PEMFE or PEFC) is a highly efficient and clean power generation system that directly converts the chemical energy of fuel and oxidant into electrical energy through an electrochemical reaction. It is the fourth type of power generation system after hydropower, thermal power, and nuclear power. Ion exchange membrane fuel cells are currently a rapidly developing next-generation fuel cell with the lowest temperature, highest energy density, fastest start-up, longest lifespan, and widest range of applications. They have promising applications in electric vehicles, submarines, portable power sources, clean distributed power stations, satellites, spacecraft, and space stations. Perfluorosulfonic acid ion exchange membranes are one of the key materials in hydrogen-oxygen fuel cells. The electrode reactants in the fuel cell can be continuously replenished, allowing the cell to operate continuously. The anode reactant is hydrogen, the cathode reactant is oxygen or air, and the battery fluid is a solid fluorine-containing ion exchange membrane.